In response to the national strategic requirements and to promote the high-quality development of the construction of high-level biological safety facilities in our country, as well as to comprehensively enhance the governance capabilities of biological safety, the Professional Committee of Pharmaceutical Environmental Engineering of the China Pharmaceutical Equipment Engineering Association successfully held the "2025 High-Level Biological Safety Facility Construction Conference" in Xiamen, Fujian Province from December 9th to 11th, 2025.

The conference venue
During the conference, the booth of Linshen Purification Group (B05) attracted many industry experts, and the on-site communication atmosphere was very lively. The company demonstrated its outstanding strength by leveraging innovative purification technologies and professional overall solutions at this grand event in the Asia-Pacific clean industry sector.
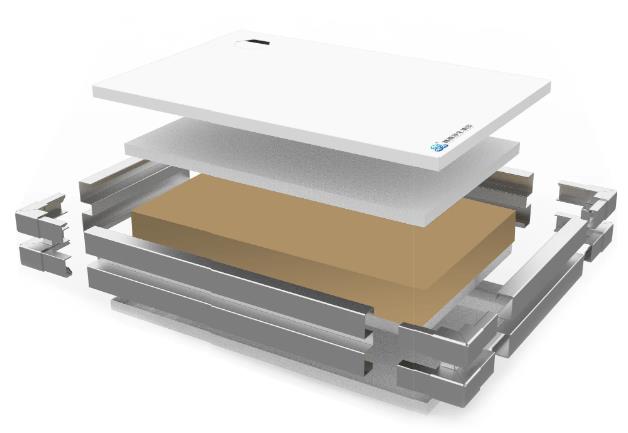

Lin Sen Purification Group Insulating Fireproof Glass / Double-Sided Glass Magnesium Rock Wool Handmade Purification Board
To address the core contradiction between "visual transparency" and "absolute security" in high-level biosafety laboratories, Lin Sen Purification Group presents two targeted solutions:
Heat-insulating fire-resistant glass,While ensuring the highest fire prevention level, achieve unobstructed observation, and enhance management efficiency and the user experience.
Double-sided glass-milled rock wool manual purification board,With superior strength and stability beyond standards, it provides a lifetime reliable airtightness and safety guarantee for the laboratory enclosure structure.
Through this conference, Linshen Purification Group not only showcased its technical accumulation in the field of cleanliness, but also expanded its cooperation opportunities and gained valuable business prospects. Linshen Purification Group will take this conference as a new opportunity to grasp the pulse of industry development, strengthen technological innovation, and work together with all partners to jointly advance, injecting new momentum into the construction of biological security facilities in our country. It will continue to delve into the clean industry and strive to become the most influential comprehensive solution provider in the global cleanroom system field.

Lin Sen Purification Group has been deeply engaged in the clean industry for over twenty years, and is committed to becoming the most influential comprehensive solution provider in the global cleanroom system field. Its core business covers: pharmaceuticals and biologics, medical devices and hospitals, biological laboratories and CMC centers, electronic wafers and new energy, food and cosmetics, aerospace and precision machinery.
The group integrates design, research and development, and product manufacturing. Relying on the CNAS-certified testing center, the product quality consistently meets international authoritative standards, leading the industry development. The group has established a rapid response mechanism (24-hour delivery support) and excellent performance capabilities for large and complex projects. Its member units operate independently and complement each other, providing customers with efficient and stable cluster services.